The AirpumpBoyle (along with his assistant Robert Hooke) discovered that sound cannot travel through a vacuum in which there is no air.
To find this he rung a bell within a hollow glass ball. It would ring normally, but when Boyle would start to pump air out of it the sound of the bell grew fainter. In the diagram of the air pump to the left, there appears to be a hand crank which allow Boyle to pump air out of the glass jar to the top. This experiment did not seem very notable at the time, however it is in fact an important moment in the history of science. He proved that there cannot be any sound without any air. Along with this he was later able to prove that all objects within a vacuum will fall at the same speed. In most situations of falling objects gravity takes over and some objects (like a ball) will fall faster than others (like a feather). |
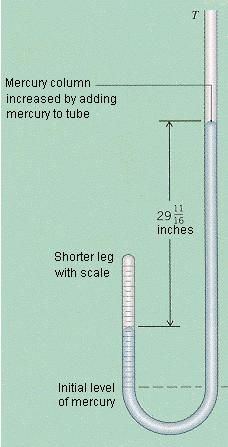
The J-tube
|
Robert Boyle's J-tube experiments lead to his ultimate discovery of the relationship between pressure and volume. Many scientists consider Boyle's experiments to be influential on modern day science because his experiments were among the first quantitative measurements performed on gas. Boyle's J tube experiment was conducted in 1662. Boyle's J tube employed the use of a J shaped glass tube, sealed on one end but open on the other (as seen in the diagram to the left). Boyle started by trapping air (acted as the gas) in the sealed side of the tube, and then he added mercury to the tube. This mercury helped to add pressure to the system. Boyle's quantitative data for this experiment was done by varying the amount of mercury (or pressure) and measured the new volume of the gas. As said in Boyle's law, this experiment was with a set amount of gas and a constant temperature. If there had been any changes in temperature the results would have thrown off Boyle's experiment. After analyzing the difference in measurements of pressure and volume, Boyle was able to determine that there is indeed a pressure-volume relationship. He measured the relationship of pressure to volume with a manometer, which he then came up with his own theory of the pressure-volume relationship. This relationship was called Boyle's law and it stated that: a gas's pressure and volume are inversely proportional in a constant environment. Boyle's J tube experiment in a short summary: - Boyle's J tube trapped air by putting in mercury - As the volume of the air decreased he added more mercury, and noted the results he saw with each change - When the volume was divided by two, the pressure doubled - This experiment lead to his law of the relationship of volumes and pressures of gas |
Proudly powered by Weebly